OVERVIEW: SMART NANOMATERIALS FOR BIOSENSING & CONTROLLING BIOLOGY
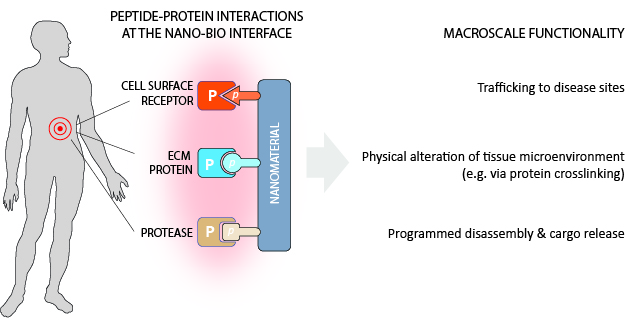
Localized changes to the tissue microenvironment during disease provide engineering opportunities for early disease detection and treatment. In particular, nanoscale materials have the necessary resolution to interface with disease biology and can be engineered to interrogate or control biological processes at the molecular scale. My work focuses on the development of smart nanomaterials that can be delivered in vivo to harness altered protease activity for disease readouts or to promote site-specific therapeutic activity. To do so, peptides are integrated into synthetic nanomaterials to modulate the interaction between engineered constructs and proteins in the diseased tissue microenvironment (i.e. cell-surface receptors, ECM proteins, proteases). At the macroscale, these simple peptide-protein interactions translate into materials with sophisticated functionalities such as (1) the ability to traffic to disease sites after in vivo administration (2) the ability to alter mechanical properties of the tissue microenvironment (for example, via crosslinking of biomolecules) and/or (3) programmed disassembly and cargo release. Using this versatile design principle, I have engineered materials to address gaps in our ability to monitor disease (such as in infectious/genetic respiratory disease) and to treat disease (such as coagulopathy after traumatic injury).